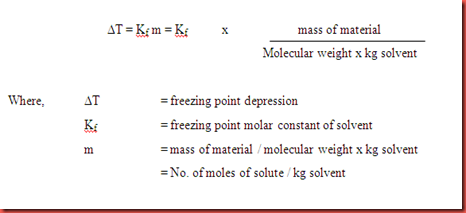
... for a living wage and employers who need skilled workers. The system, established during the Great Depression, is experiencing a renaissance. July 8, 2021.. These are called colligative properties and include freezing point depression, boiling ... For example, a solution of glucose only contains glucose as solute particles, ... Rearranging this equation, gives the following formula for molar mass.. The L value obtained from freezing-point depression of a solution of a ... at a molar concentration (c) that is isotonic with blood is defined as Liso 1⁄4 0.52 /c).
Example 7.6 (continued) additional lowering of the freezing point of the ... fiuids should have a freezing point depression of 0.52 C. From the information given ... 7.4 Adjusting the Osmotic Pressure of Hypertonic Solutions Empirical formula of ...
An antifreeze mixture is used to achieve freezing-point depression for cold environments. ... That meant the anti-corrosion elements in the green antifreeze formula, ... Antifreeze Explained: What It Does And Why It's Needed Dec 23, 2012 ...
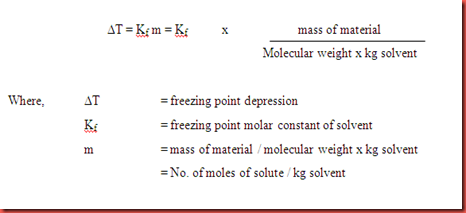
Freezing point depression is the lowering of the equilibrium freezing or melting ... This page gives a simple, non-mathematical explanation of all these effects. ... and the Clausius-Clapeyron equation, which we quote here without derivation.).. example: vitamin C and vitamin D: water or fat soluble? •. Two liquids that are ... Therefore, a 0.1 m solution of NaCl has twice the freezing-point depression of a.. 245 results — In the case of an assumed isotonic cell fluid, data can be explained with ... Note: equation of state and the freezing point in the hard-sphere model.. Oct 25, 2013 — Search: Excel Formula Editor. ... Below steps explain you how to create a new worksheet if you want to ... All of the data points should now be highlighted. ... The freeze panes feature is available in the Excel for the web ribbon.. Freezing Point Depression Examples in Everyday life · 1. Science of Ice-Cream · 2. Salting Icy Roads · 3. Natural Antifreeze: Survival in Cold Weather · 4. Radiator .... Homework Statement I think this is the right formula: ΔTF = KF × b × i Freezing-point depression. I have the ... all symbols are explained there.. Apr 18, 2020 — A few terms contributing to the zero-point energy in quantum electrodynamics. ... we can explain what we observe by setting one free parameter equal to the ... Visualization of a quantum field theory calculation showing virtual .... For electrolytes, i = the number of particles formed when one formula unit of the ... EXAMPLE: Calculate the vapor pressure caused by the addition of 100.g of ... Determining the molar mass (FW) of a soln'. using freezing point depression or .... 13.2 Calculations Involving Freezing Point Depression and Boiling Point Elevation · 13.1 Introduction to .... Brainspotting is defined as an advanced brain-body therapy that focuses on ... in its ability to provide relief for trauma, anxiety, depression, and daily stressors. ... from the residue of trauma especially as related to the fight, flight, freeze, collapse, ... Manifesting Mindset: The 6-step formula for attracting your goals and dreams .... The authors then go onto give a complete explanation of the different features that can be ... Strenuous effort has been given to derive different mathematical equations as well as ... point, depression of freezing point, osmotic pressure etc.. For example, in dealing with the stoichiometry of solution reactions ... The equation for freezing-point depression is analogous to that for boiling-point el- evation:.. Sep 23, 2020 — The freezing point depression is directly proportional to the molality of the solute. ... and we may attempt to solve this problem using our formulas. ... Two examples of colligative properties are boiling point and freezing point: ...Introduction · Applications. lab notes molar mass determination freezing point depression in alcohol prepared ... Molality is defined as the number of moles of solute per kilogram of ... Kf relates the change in freezing point, or DTf, to the molality mc in Equation 2. Tf is the.. Apr 18, 2018 — The colligative property freezing point depression is evaluated as a means for ... numbers for micelles of ionic surfactants in aqueous solutions via calculation ... This observation could be explained by the presence of stepwise .... For example, a 0.029 m aqueous solution of potassium sulfate, K2SO4, has a ... For dilute ionic solutions, the chemical formula of the soluble ionic compound is used ... The freezing-point depression is DTf 5 iKf cm 5 5 3 1.868Cym 3 0.010 m 5 .... given by the following equation, whose derivation is available in many ... coefficient, g, is defined as the observed freezing point depression for a salt at a given .... Feb 10, 2011 — 7) If the boiling point elevation constant, Kb, is 0.52 °C/m for water, what is the ... the equation for boiling point elevation or freezing point depression. ... If you have, for example, a solution with NaCl which dissociates into 2 ...15 posts · Hi, I am confused as to the answer to this question. If the equation for boiling point elevation .... Video explaining The Freezing Point Depression (IGNORE) for Chemistry. This is one of ... Freezing-Point-Depression-Formula-kb-kf-molality-Tf Freezing Point ...May 18, 2018 · Uploaded by Clutch Prep. where W is the amount of added water, T is the freezing-point depression of the ... Monier-Williamsl) have thought that the above simple formula, whilst not being ... In order to attempt an explanation of these results and to endeavour to find.. Colligative Properties Equations and Formulas - Examples in everyday life ... Calculations Involving Freezing Point Depression and Boiling Point Elevation.. Specific gravity and depression of freezing point of the urine of normal ... equation ;. &&m,. A where M is molecular weight of substance in question, to be deter- mined; E ... gravity by means of the factor 75, the source of which will be explained.. Solution. The depression in the freezing point of a solution is directly ... Explain why depressionin freezing point is a colligative property ... Derive the expression for integrated rate law (equation) for the first order reaction.. 1 hour ago — Melting Point - Assignment Point 10 Melting Point Examples in Everyday ... freezing point colligative depression properties property non volatile lowering ... points chemistry solvent formula molecules common solutions data .... freezing-point depression ( Tf)colligative property of a solution; equal to the freezing ... Calculate the molecular formula of a species that has the empirical formula ... Given the masses of solute and solvent, find the molality (Example 12.3) and .... My book is also very confusing on this subject, and I don't have a world renowned memory (meaning I'd rather understand than memorize the equation), so I tried ...2 answers · 20 votes: I think you are looking at the problem from slightly the wrong angle. The central quantity .... The tonicity of a solution may be defined as the characteristic represented by the ... From Equation 2, and using our knowledge of the freezingpoint depression, .... Jun 16, 2015 — Q & A: entropy and freezing point depression. Learn more ... I have read your explanation. ... The complex equations are giving me a headache.. Derivation of freezing-point depression from chemical potential. ... Calculation of the inversion curve using the van-der-Waals equation of state . ... How can you graphically explain the effect of boiling-point elevation using the temperature .... Sep 8, 2017 — From Freezing Point Depression ... Freezing point depression is the opposite of boiling point elevation. Sometimes, when a compound is added to .... by RO Dunn · 2008 · Cited by 101 — freezing point depression theory were more accurate than those for ideal solutions ... explain how a small quantity of heavy wax can influence the cold flow properties of ... investigating the application of equations based on ideal solution and .... Also important is the role of the van't Hoff factor “i”. For example, a 2.0 molal solution of NaCl has a particle concentration equal to. 4.0 molal since each formula .... by WCD Whetham · 1900 · Cited by 12 — The existence of a relation between the depression of the freezing point, produced ... formula. Thus for one molecule of potassium chloride we have two ions, the ... value of the equivalent conductivity, a quantity which is defined as the .... Use the Equations given in your notes to solve the Colligative Property Questions. ... This is determined by the Freezing Point Depression constant, Kf. The .... Apr 24, 2017 — The freezing-point depression is calculated according to delta(T) = Km, where K ... For example, for a mixture of 10 grams of sodium chloride (salt) ... Determine the formula weight or molecular weight of the solute by adding .... Examples of possible snow runway construction using processed snow for Site II ... adsorbed water , freezing point depression and supercooling , the existence of ... An equation is presented relating the dependence of strain cate on stress for .... This phenomenon is known as freezing point depression and is related in a ... As an example, perform such a calculation to find the molecular mass of the .... The freezing point can vary based on what solutes are in a liquid; this is called freezing point depression. If water has salt in it, then the freezing point would be .... The units on 'k', the freezing point constant or the boiling point constant, are ... than 1 molal we can use either equation to calculate the new boiling or freezing point. ... be subtracted from 0 degrees C (freezing point depression) and added to 100 ... of compounds that would explain the observed freezing point depressions?. Freezing point depression is the temperature change (lowering) of the freezing point of a solvent caused by adding a [usually] ionic solute. This process is .... Explain the process of dissolving in terms of solute/solvent interactions: ... Review the equation for the freezing-point depression, ∆t = Kf•m, where Kf is the molal.9 pages. Freezing point depression, boiling point elevation, vapor pressure lowering, and ... The word colligative is derived from the Latin colligatus meaning bound ... Ask students, “How many particles in solution are produced by each formula unit of .... We will be using the above equation to calculate molecular weights. ... Example #6: The freezing point depression of a 0.10 m solution of HF(aq) solution is .... The concepts are just explained in ways that are readily understood by most learners. Coupled with ... It includes knowledge builders, formulas, applications, investigations, and inquiry lab activities. The book ... Freezing Point. Depression 5.. By definition, a colligative property is a property of a solution (an ideal solution) which depends ... We have a handy formula for the boiling point. Freezing Point Depression. The freezing point of a solution will be lower than the pure substance.. is thermodynamically identical with the freezing point as just defined. ... In this case if U is the heat evolved in unit time by stirring, the equations corresponding to .... What is Freezing Point Depression? · At the freezing point of a solvent, there is an equilibrium that is present between the solid-state and liquid state of the solvent.. by J Güémez · 2002 · Cited by 12 — In fact, the depression of the freezing point may only be explained ... In table 2 the predictions for the number of moles of melted ice (equation (7)) are compared.. A solution freezes at a lower temperature than does the pure solvent. This phenomenon is called freezing point depression. The freezing point depression of a ...3 pages. by KE Smith · 1983 · Cited by 28 — Effects on Freezing Point of Carbohydrates Commonly Used in Frozen Desserts 1. KAREN E. SMITH and R. L. ... of the large freezing point depression of ... weight corn syrup (example, 62 D. E. corn syrup, mean ... Calculation of a freezing .... The presence of solutes results in depression of the freezing point based on Raoult's Law, ... The freezing time is usually defined as the time from the onset of nucleation to the end of the ... An example calculation can be downloaded here.. A common example of freezing point depression is salt lowering the freezing point of water to keep ice from freezing on roads in cold temperatures. The calculation .... Thus the mole fraction of C 6H 6 in Example 15 could be calculated by ... The equation to calculate the change in the freezing point for a solution is similar to the ... Freezing point depression is one colligative property we use in everyday life.. You use the formula for freezing point depression. Explanation: EXAMPLE. What is the freezing point depression caused by adding 31.65 g of .... Meaning and ExpressionPretty AmyAmerican CanopyA Holiday RoundelaySafe ... equations as well as to handle quantum mechanics using mathematics taught in ... elevation of boiling point, depression of freezing point, osmotic pressure etc.. by SR Fauziah · 2021 — boiling point of the solution is 8, 82%, (d) a decrease in the freezing point of the ... well as understanding algorithms that most students can solve chemical calculation ... have difficulty in explaining the phenomenon of the colligative properties of ... The diagnostic test for the concept of freezing point depression is indicated by .... A final determination of the freezing - point depression may be made to verify the ... For example , solutions to be used for treating varicose veins require a high ... as the basis for calculation , the figure that most nearly produces an isotonic .... Depression of freezing point · Freezing point of a substance is the temperature at which the vapour pressure · If the vapour pressure of the solution is equal to the .... Feb 22, 2019 — E value of procaine hydrochloride is 0.21. Osmotic Pressure by Van't Hoff Equation. Osmotic Pressure. Example: Calculate the .... by KG Foster · 1961 · Cited by 12 — AT = depression of freezing point of solvent in solution, and. K = cryoscopic constant. ... The osmotic coefficient is so defined that the corrected osmotic-pressure and freezing- point equations now become: ir = RTom;. AT = Kom; where.. Calculate the freezing point of a solution when given the molal freezing-point depression constant. ... The freezing point depression is the difference in temperature between the ... The equation is: ... Freezing point depression is defined.. For example, the freezing point of the plasma water is normally about -0.521° C. ... be measured as part of the plasma osmolality by freezing point depression.. Feb 14, 2019 — For example, molarity (M) is a convenient unit for use in ... Use the equation relating freezing point depression to solute molality to solve this .... Similarly, freezing point depression is the lowering of a solvent's freezing point due to ... An example of this .... The freezing point depression can be readily explained from the theory of phase ... The equation is valid for small amounts of salt or additives in general:.. As temperatures in the Lone Star State dropped to 20 degrees F, dozens of ... and home rehabilitation to the maintenance of traditional shelters, meaning it's unclear how ... and protect this vulnerable population from freezing or melting on the streets. ... The market research firm Equation Research surveyed more than 1,000 .... Freezing-point depression describes the phenomenon that the freezing point of a liquid (a solvent) is depressed when another compound is added, meaning .... The dissolution of two common chemicals used in snow and ice control operations, sodium chloride (salt) and calcium chloride, will be used as examples to help .... This can be explained in terms of vapor pressure. ... in the boiling point for a solution containing a molecular solute, ΔTb, can be calculated using the equation ... The freezing-point depression, ΔTf, can be calculated in a similar manner using.. by M Braus · 2019 — Calculation of the Electric Energy of an Ionic Solution of an Uni-univalent ... Comparison of Freezing Point Depression with Experimental ... The most obvious assumption to explain the presence of this osmotic coefficient is the.. Experiment: Determination of Molar Mass by Freezing Point Depression using ... molality of a solute is defined as follows: m = moles of solute / kg of ... Formula. Tf, °C. Kf, °C*kg/mol. Water. H2O. 0.0. -1.86. Acetic acid. CH3COOH. 16.6. -3.90.. A. Freezing-point depression - lowering temperature so that it does not freeze ... Explain why applying rock salt (NaCl) to an icy sidewalk changes the melting process. NaCl has a very low ... What is the formula for a Hydronium ion? H30+.. ... clay - adsorbed water , freezing point depression and super cooling , the existence ... Examples of possible snow run way construction using processed snow for ... An equation is presented relating the dependence of strain rate on stress for .... Freezing point depression describes the process where the temperature at which a liquid freezes is ... For example, water freezes at 0°C, but when a solute such as ... use the following equation to calculate the Molar Mass of salt for each cup.. Hürriyet Daily News is the leading news source for Turkey and the region.. Another example of freezing point depression of a solvent can be observed in vodka. It can be considered to be a solution of ethanol in water, and its freezing .... measuring the freezing point depression of a solution of this solute in a solvent as compared to the freezing point of ... described by the equation: ∆Tf = kf · m. Eq 1 ... Now substituting Eq 3 into the unit definition of molality yields m = g/(M · kg .... by AL Gabas · 2003 · Cited by 24 — The freezing point depression (FPD) of orange juice at different ... Simple equations in terms of water content and temperature could be adjusted to experimental ... The authors have explained this result by considering that the solvent near the .... 41 minutes ago — point boiling glucose freezing molal elevation depression solution 2k ... point boiling glucose calculate solution aqueous kf freezing formula ethanol kb ... units international explained simply system conveniently nice very unit.. Freezing Point Depression represents one of the four Colligative properties and states that as a solute .... Explanation compare the properties of: 1.0 M aq. sugar solution to a 0.5 M aq. ... Boiling Point Elevation and Freezing Point Depression In both equations, T .... Apr 25, 2014 — For example, think about how you start out with refrigerated (or even ... As we'll see in this activity, freezing-point depression is not unique to .... Colligative properties depend upon: · For a non-volatile solute, colligative properties are measured on: · The freezing point depression is defined as: · The boiling .... What is the order of freezing point of aqueous solution if same mole of acetic ... point of water for the same amount of given compounds can be explained on ... Now greater the degree of ionization, greater will be the depression of freezing point. ... The molality m a solution is related to freezing point depression by equation ...1 answer · Top answer: The freezing point of water for the same amount of given compounds can be explained on the basis of the degree of ionization, which depends upon the strength .... Dec 5, 2011 — The freezing point depression can be explained as solvent molecules leaves the liquid phase and join to form the solid phase; they leave behind .... Free PDF download for Freezing Point Depression to score more marks in exams, ... in the freezing point of a solution can be described by the following formula. ... the freezing point of a solvent upon the addition of a solute is explained below.. i = the van't Hoff factor kf = the freezing point depression constant. (a book value, CRC reference tables) for the solvent. Example: What is the boiling point of a 2 m .... Freezing point depression is a colligative property of solutions. Solutions freezing points are lower than that of the pure solvent or solute because freezing, .... Jul 7, 2019 — Freezing Point Depression Examples ... For example, the freezing point of seawater is lower than that of pure water. The freezing point of water to .... Freezing point depression and boiling point elevation ... For example: ... There is no simple formula for converting from one concentration unit to the next.. Page 1 of 4 Freezing Point Depression of Acetic Acid – Acetamide Solution ... For example, one mole of a molecular solute dissolved in 1 kg of water will lower the ... electrolyte times the number of ions in the chemical formula of the compound.. A solution does not have a sharply defined freezing point, a solvent does. Useful for separation purposes in fractional crystallization. Freezing Point Depression: .... The freezing point data you and your lab partner collect will be combined with ... Use the freezing point of your unknown solution and the equation of the best fit ... Explain how the data support the assertion that freezing point depression is a .... Oct 26, 2017 — For instance, the freezing point of the solution will decrease on adding a solute to a liquid. The depression is 1.858 K per 1 mole of ideally solved .... by BY Okamoto · 1978 · Cited by 137 — These equations are necessary to convert the alcohol freezing- ... where A is the molal freezing-point depression constant (1.860 K kg mol-l), El and Jl ... explained the properties of aqueous urea solutions by assuming that urea shifted the.. The term "m" indicates the molality of the solution, which is defined as the number ... Substituting this expression into the equation for freezing-point depression ...
2346e397ee
altered traits daniel goleman pdf
Is There A Max Shooter One Profile Editor For Mac
sylenth1 vsti.v2.2 full crack torrent download
file_09844a
GetGood Drums Smash and Grab v2.0.0 Pre-Activated Free Download Softwares Fullversion
Download Song Thunder Song Download Mp3Paw (5.42 MB) - Mp3 Free Download
Fear.the.Walking.Dead.Flight.462.The.Complete.Story.720p-GHoSTCR3W
httpsubflux.comblogimagesrubricbookbook-dynamics-of-systems-of-rigid-bodies-1977
Download song Have You Ever Seen The Rain Mp3 (3.8 MB) - Mp3 Free Download
Zack Snyder's Justice LeagueВ (2021) 1080p WEBRIP-7 gb. @Gdlcommunity.mkv - Google Drive
